Red Blood Cell–Mediated mRNA Delivery for Improved CAR T Cell Therapy for Cancer
Gene therapy is taking a new dimension each day, bringing new possibilities for treating diseases. A new major research has been accomplished in the area of cancer research. The study offers the potential to alter the future of CAR T cell therapy by providing a quicker and easier process for making an individual’s own immune system a very effective weapon against cancer. Researchers have developed a delivery method that uses red blood cells (RBCs) as carriers of genetic instructions, delivering them directly into the body. This may allow the elimination of all complex laboratory procedures that define today’s CAR T-cell method.
The results from a new study published in Science Translational Medicine present a completely new approach to introducing the modified genetic material into T-cells. In that, RBCs are used to carry messenger RNA (mRNA) containing modified genetic material to reprogram T-cells to recognize and attack cancer. This new approach does not require that T-cells be taken from a patient, sent to a laboratory for processing, and then returned to the patient weeks later, as in current CAR T-cell therapy.
Why RBCs? Rethinking the Role of Red Blood Cells
Red blood cells are abundant and circulate oxygen throughout the body without eliciting a strong immune response. Now, the researchers are exploring a new dimension by using them as delivery vehicles for therapeutic purposes. As they are present naturally and are less likely to trigger the immune system, they can be used as vectors, unlike synthetic or viral vectors.
Red blood cell characteristics enable efficient mRNA delivery and mask mRNA from detection by immune surveillance mechanisms, which have been limited in their efficacy to date in improving CAR T-based therapies.
How Scientists Engineered These Cells
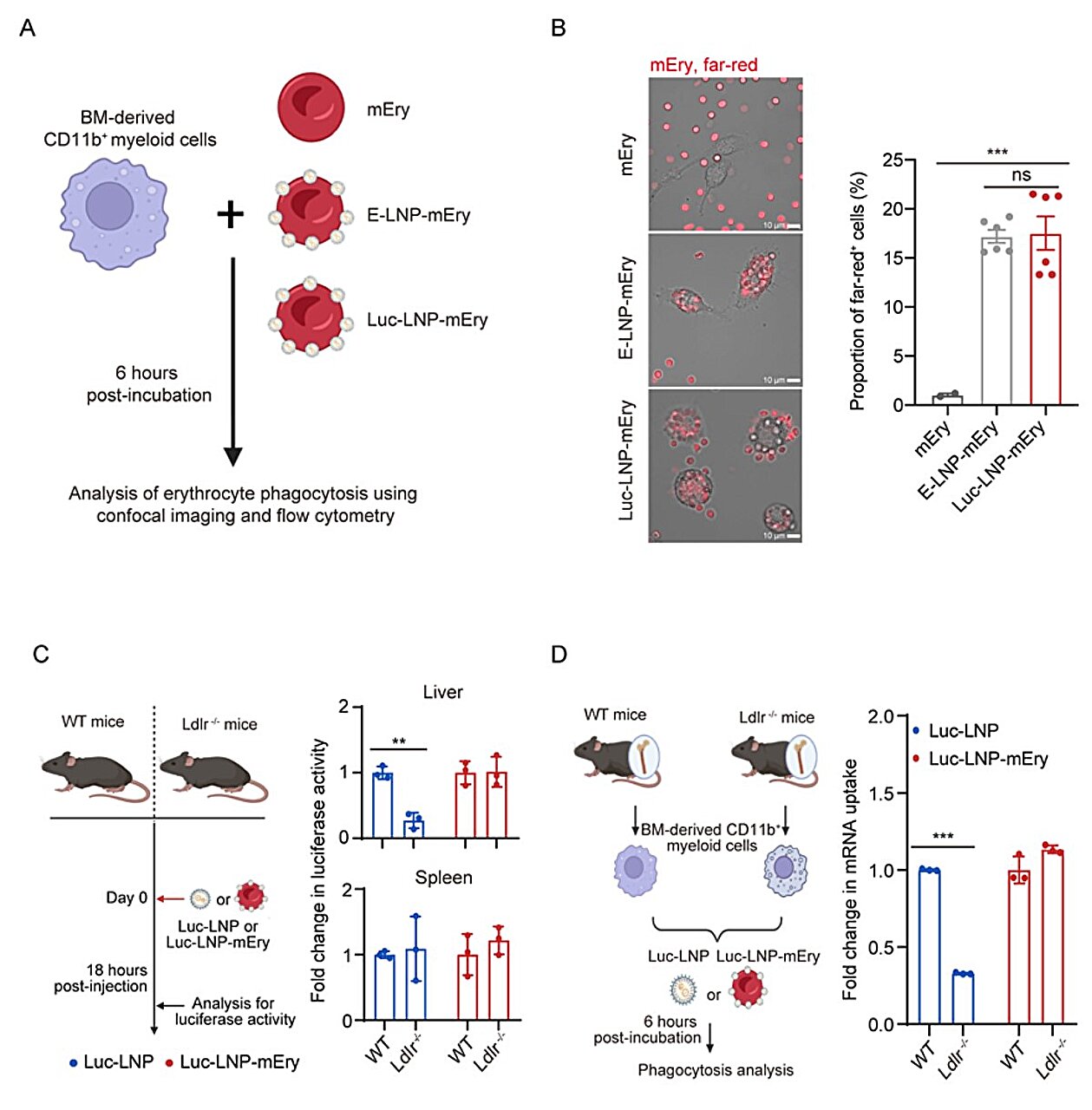
In this study, researchers are using erythrocytes, or red blood cells, as drug delivery systems. The researchers developed an erythrocyte-mediated messenger RNA delivery platform, called mRNA-LNP-Ery, in which mRNA-loaded lipid nanoparticles are covalently bound to erythrocytes.
As Dr. Xiaoqian Nie, lead author of the investigation, said, this technology holds great therapeutic promise, as mRNA-LNP-Ery can travel freely throughout the human body by circulating in the bloodstream. Red blood cells transport lipid nanoparticles that carry mRNA encoding genetic information to activate myeloid cells (such as macrophages) in the patient’s body, turning those cells into cancer-targeting agents.
This in vivo programming of myeloid cells represents a substantial shift from the current CAR T cell treatments, which utilize ex vivo methods of programming cells, making CAR T treatment faster to deliver and potentially scalable.
Conjugated LNPs mediate splenic recognition and uptake of LNP-Ery. Credit: Science Translational Medicine (2026). DOI: 10.1126/scitranslmed.ady6730
Advantages over Traditional CAR T Cell Therapy
The potential impact of this new method of delivering CAR T cells to patients is particularly relevant in overcoming significant barriers present in the current CAR T therapies.
- No longer need to extract or modify cells for the infusion period
- Significantly less time required to deliver treatment – potentially reduced from weeks to days
- Overall treatment costs should be significantly lower
- Expand access to treatments that are currently limited to specialized centers.
This innovation will ultimately help to expand the availability of CAR T cell therapies to patients who cannot obtain CAR T therapy because they cannot access treatment as a result of current barriers to the delivery of CAR T cells to patients.
Expanding Beyond T Cells
Another important shift is the type of immune cells being targeted. While CAR T cell therapy primarily focuses on T cells, this new technique reprograms myeloid cells, which are naturally better at infiltrating solid tumors.
In preclinical studies, these engineered cells:
- Migrated directly to tumor sites
- Destroyed cancer cells effectively
- Boosted the activity of T cells and natural killer cells
This implies that the strategy would provide complementary or enhancing effects to CAR T cell therapies when used to treat solid tumors, for which current therapeutic options are not considered successful.
Challenges and Ongoing Research
Although this research is still preclinical, the potential implications for patient care are tremendous. If clinical/ healthcare studies yield successful human trial results, this opens a new door to CAR T cell therapy delivery by providing faster, less expensive methods of administration that will allow a larger number of patients to benefit from CAR T therapy.
This discovery is not designed to replace traditional CAR T cell therapies; instead, it will create a new phase in the evolution of CAR T cell therapies. By combining technology-based genetic engineering of the patient’s own cancerous cells with the natural delivery mechanism of red blood cells, scientists hope to create a future in which cancer therapies. This can be deployed at scale and delivered more easily to patients, while offering improved outcomes over currently available options.