US Military to develop “Genetic Doomsday” Weapon to wipe Entire Species
Our understanding of the natural world is now so great we can manipulate the DNA blueprints for any living thing on Earth. We can replace genes for traits we don’t like with others we prefer and even add genes that don’t occur naturally in an organism. And in 2013, scientists discovered a new way to precisely edit genes — technology called CRISPR that further raised all sorts of enticing possibilities.
One of the more intriguing ideas came from Kevin M. Esvelt and his colleagues at Harvard University: Crispr, they suggested, could be used to save endangered wildlife from extinction by implanting a fertility-reducing gene in invasive animals — a so-called gene drive.
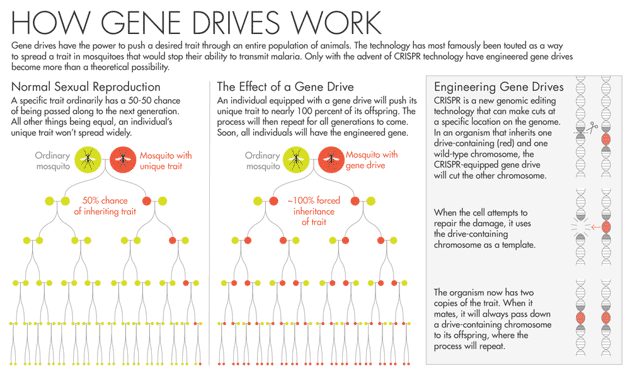
Gene-editing techniques involving cutting genes at specific sites in the DNA of an embryo in order to disrupt those genes’ function or insert other genes. But there are other ways gene-editing has the potential to help in the fight against infectious diseases. This process is referred to as a “gene drive”, and furthermore, isn’t new: nature spreads evolutionary changes through a population all the time. It doesn’t mean changing the DNA of all living individuals in a population. Instead it’s about ensuring a specific genotype (a certain version of a gene) is passed on to the descendants of modified individuals.

A sexually reproducing organism usually has a 50% chance of inheriting a specific genotype from one of its parents. Using a gene drive can bias the inheritance pattern to increase that chance to nearly 100%, ensuring almost all descendants possess the genotype. As those descendants mate and produce their own offspring, the proportion of organisms with the genotype increases until it can be found in the entire population.
Besides the idea that you can “replace” a population’s genotype is particularly appealing when that population is responsible for spreading disease. And the US military’s research arm is among one of the technology’s biggest research funders. It’s no secret that the Defense Advanced Research Projects Agency has its eye on gene drive technology.
DARPA’s interest in gene drives—and what exactly the sometimes secretive agency plans to do with them—is making headlines again this week after an anti-gene drive advocacy group published a trove of emails from gene drive researchers obtained through a Freedom of Information Act request.
In July last year, Darpa awarded $65 million in four-year contracts to seven teams of scientists to study gene-editing technologies. The commitment officially made Darpa the world’s largest government funder of gene drive research. Most of that money is going toward designing safer systems and developing tools to counter rogue gene drives that might get into the environment either by accident, or with malicious intent.
Darpa is investing $100m in genetic extinction technologies that could wipe out malarial mosquitoes, invasive rodents or other species, emails show.
More specifically, the agency is investing this money into technologies which can wipe out dangerous animals and insects. The first order of business is getting rid of malarial mosquitoes, although invasive rodents and other undisclosed species are also on the list.
To date, there have been no synthetic gene drives released on wild populations, although there is an increasing number of papers investigating their use, specifically when it comes to eradicating malaria-carrying mosquitos. This is how many have imagined it being used, but even then the ecological impacts of such widespread genetic manipulation of wild creatures are still not understood.
The seven teams that DARPA is funding under its Safe Genes program are primarily conducting fundamental research. A team at The Broad Institute is developing the means to switch on and off genome editing, including the control of gene drives in mosquitos. A Massachusetts General Hospital team is looking at better ways to measure the on-target and off-target effects of gene editing, with a focus also on a mosquito gene drive. An MIT team is looking at how to geographically limit the spread of a gene drive and potentially reverse it. Five of the funded teams are working directly on biosafety and biosecurity measures for gene drive technologies, the other teams focused on gene editing more broadly.
The concern, of course, is that a military organization like DARPA could turn around and take those those technologies for offensive measures—maybe, say, a gene drive that destroys an enemy’s crops. It’s potentially scary technology. It’s easy to let the imagination run wild.
Potential military applications for this research are also of concern, although none seem to exist right now. The US military is always looking for new tools, and genetic extinction technologies can always be weaponized in one way or another. DARPA doesn’t have the best of reputations in this regard either, which will only make matters worse.
Unintended consequences are always difficult to predict, especially when these technologies are tested in the real world. For example, the mosquito population may no longer spread malaria, but it may affect other species along the way. There is a lot we don’t know yet about how nature works, and messing with things one doesn’t understand is never a good idea.
What is certainly clear from the documents, though, is that DARPA has a hand in most of the significant research into gene drives in the country. And that the military takes very seriously the potential for a world-altering technology to become a serious threat.