A Breakthrough In Plant Science Using Optogenetics For Plant Research
It is practically a decade now after the release of the scientific journal namely optogenetics which is referred to as the breakthrough of the decade. In simple words, the methodology facilitates controlling the electrical functioning of the cells using light pulses. With its aid, researchers can perform novel discoveries and acquire an in-depth understanding of the metabolism of nerve cells, for instance, and hence better perceive psychiatric & neurological diseases like schizophrenia & depression.
Established procedure on animal cells
Optogenetics has now become an established methodology employed in several sectors especially in research related to animal cells. The technique is different in plant studies: shifting the principle to plant cells & implementing it extensively has not achieved completely till now.
Nevertheless, this has currently altered: Researchers from the JMU (Julius Maximilians University of Würzburg) have successfully applied optogenetic techniques in tobacco plants. They provided the results of their study in the latest release from the journal, Nature Plants. Prof. Georg Nagel, optogenetics co-founder stated that Dr. Shiqiang Geo belonging to his team, and Dr. Kai Konrad belonging to Prof. Hedrich’s team (Botany I) were particularly responsible for this achievement. Apart from the Department of Neurophysiology, Institute of Physiology, 3 chairs from Julius-von-Sachs Institute were present in the partnered study: Pharmaceutical Biology and Botany I & II.
The light switch for cell activity
Nagel explained the co-developed method, that optogenetics is the tailoring of a living organism or their cells using light by introducing a light sensor into them with genetic engineering techniques. Especially, the light-regulated cation channel, channelrhodopsin-2, has aided optogenetics in achieving the glorious victory. Along with channelrhodopsin, the cell activity can be turned off & on like a light switch.
Within plant cells, nonetheless, this has till now just operated to a limited level. Dr. Gao elucidated that there are 2 primary causes behind this: It is hard to genetically tailor plants in such a way that they normally produce rhodopsin. Also, they don’t possess an important cofactor. Without this co-factor, rhodopsins can’t function- all-trans-retinal, which is also referred to as Vitamin A.
Greenlight for plant cells
Dr. Konrad, Dr. Gao, Prof. Nagel & other teammates have currently been able to resolve both issues. They were able to successfully produce Vitamin A from tobacco plants by the introduction of an enzyme isolated from a marine bacterium, thereby facilitating an ameliorated integration of rhodopsin into the plasma membrane. This enables, for the very first time, non-invasive engineering of intact plants or chosen cells using light mediated by the anion channelrhodopsin, GtACR1.
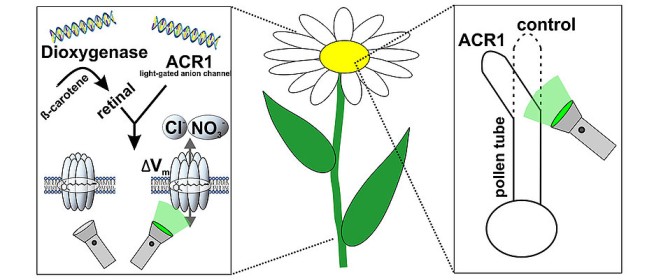
In a previous technique, Botany I-plant physiologists had artificially incorporated the much-needed Vitamin A co-factor into the cells to facilitate a light-fenced cation channel to transform into an active one within plant cells. Employing the genetic engineering now presented, Prof. Nagel & the team have produced plants that generate a particular enzyme along with rhodopsin, known as dioxygenase. These plants are later able to generate Vitamin A-which is normally absent within plants-from provitamin A which is rich in chloroplasts of plants. The blend of rhodopsin optimization & Vitamin A production for plant application finally directed the scientists supervised by Dr. Konrad & Prof. Nagel to victory.
A new approach for plant science
Dr. Konrad claimed that if these cells are irradiated using green light, the cell’s permeability for negatively charged entities increases substantially, and the membrane potential is altered drastically. Using this approach, he added that it is feasible to particularly engineer the pollen tube growth & leaf development, for instance, and thereby to explore the molecular dynamics of the plant growth process explicitly. The Würzburg scientists are certain that this unique optogenetic method to plant research will substantially help with the analysis of previously misconstrued signaling paths in the future.
A pioneer of optogenetics in plant research
Rhodopsin is a light-sensitive plant pigment that forms the foundation of vision in numerous living beings. It is also present naturally. The concept that a light-sensitive ion transport channel isolated from an archaebacterium (bacteriorhodopsin) can be added to vertebrate cells and operate was initially illustrated by George Nagel in the year 1995 along with Ernst Bamberg from the Max Planck Insitute for Biophysics, Frankfurt. In 2002/2003, this evidence was parallel succeeded with light-sensitive ion channels isolated from algae.
Along with Peter Hegemann, Prof. Nagel explained the continuance of the two light-sensitive channel proteins, ChR1/ChR2 (channelrhodopsin-1 & 2), in two articles released in 2002 & 2003. Importantly, the scientists found that ChR2 activates a really fast, light-induced alteration in membrane voltage & current when the gene is translated in vertebrate cells. Also, ChR2’s miniature size provides convenience.
Prof. Nagel has from then acquired several awards for this breakthrough, most lately in 2020-along with 2 other discoverers of optogenetics-the Shaw Prize for Life Sciences ($1.2 million).
Plant science, Optogenetics In Plant Research























