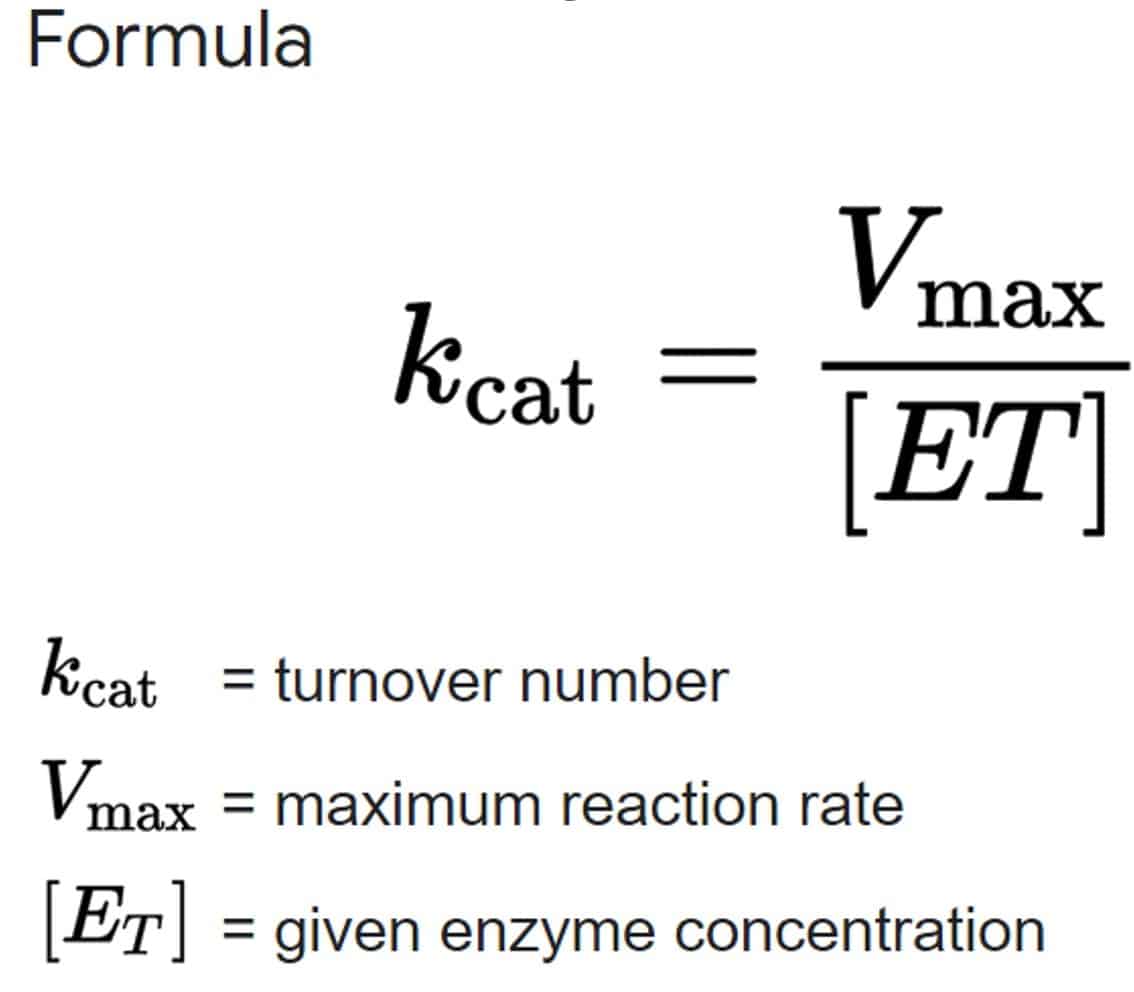CSIR Enzyme Kinetics Notes + Flowchart Download
The Topic Enzyme Kinetics belongs to CSIR NET UNIT 1: Molecules and their Interaction Relevant to Biology. You must not skip this unit as it has a connection with other units mentioned in CSIR NET Syllabus. For eg: proteins, you find in almost all other units. If you skip this UNIT you will find difficulty with concepts mentioned in other units. No doubt, it is the 1st unit of the syllabus!
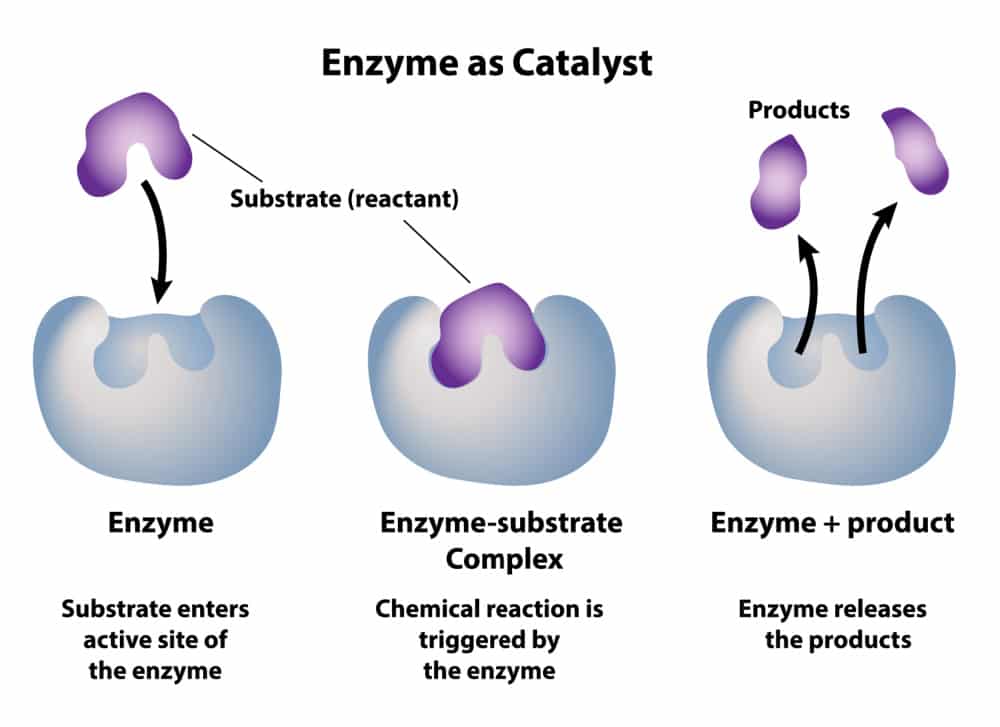
Enzyme:
- Globular protein which functions as a biological catalyst
- Speeds up the reaction rate
- Lowers activation energy without being affected by the reaction it catalyzes
- They catalyze nearly all the chemical reactions taking place in the cells of the body
- Have unique 3D shapes that complementary fit the shapes of reactants (substrates)
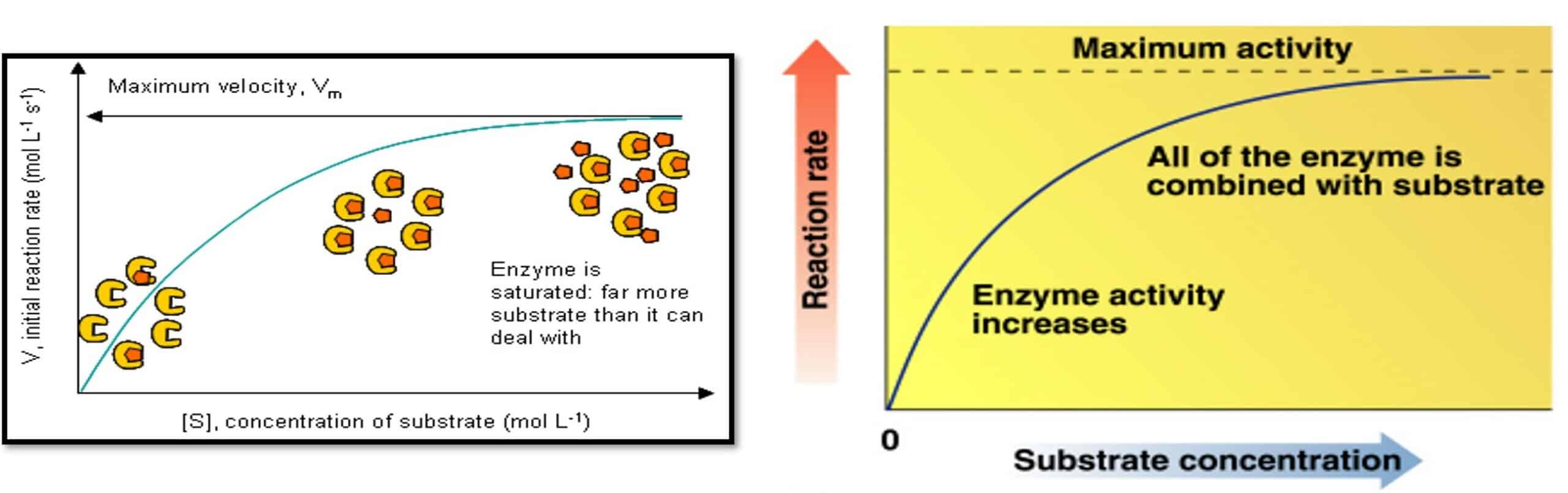
Substrate Concentration and Reaction Rate
- The rate of reaction increases as substrate concentration increases (at constant enzyme concentration)
- Maximum activity occurs when the enzyme is saturated (when all enzymes are binding substrate)
Substrate concentration: Enzymatic reactions
- Faster reaction but it reaches a saturation point when all the enzyme molecules are occupied.
- If you alter the concentration of the enzyme then Vmax will change too
Effect of Substrate Concentration on Reaction Rate
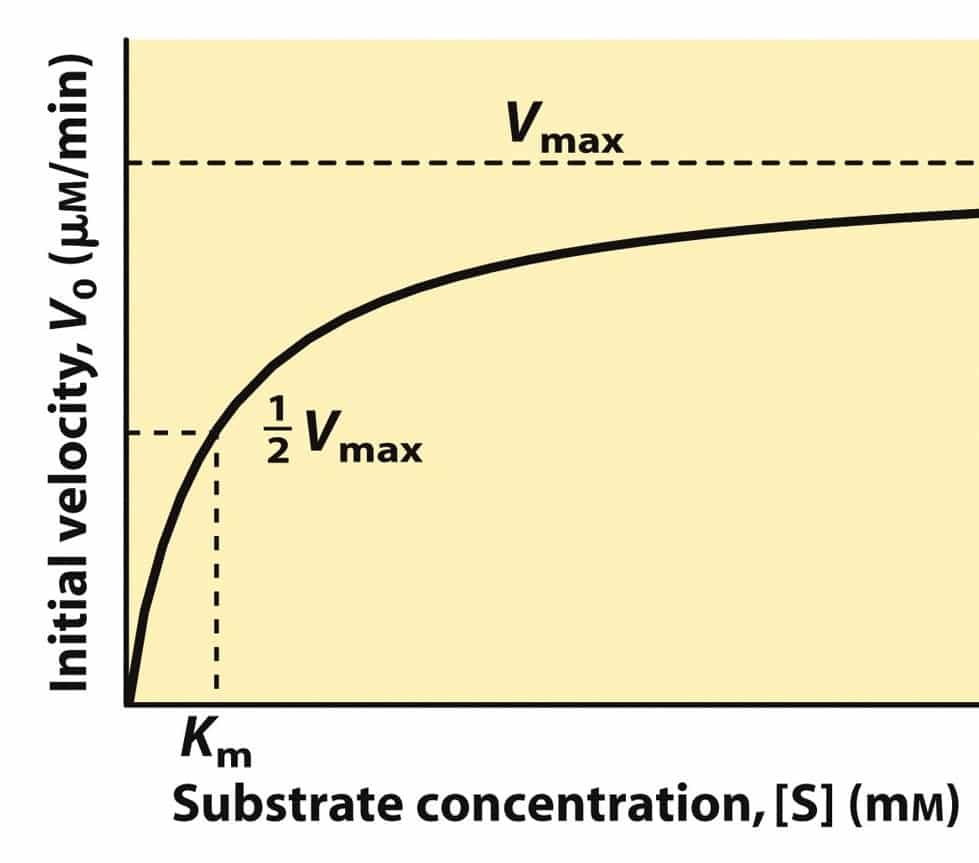
- Measurement of velocity:
- V = rate of appearance of product = change in [P] per unit time
- The initial velocity increases with [S] at low [S].
- [velocity =d[P]/dt, P=product]
- The initial velocity approaches a maximum at high [S]
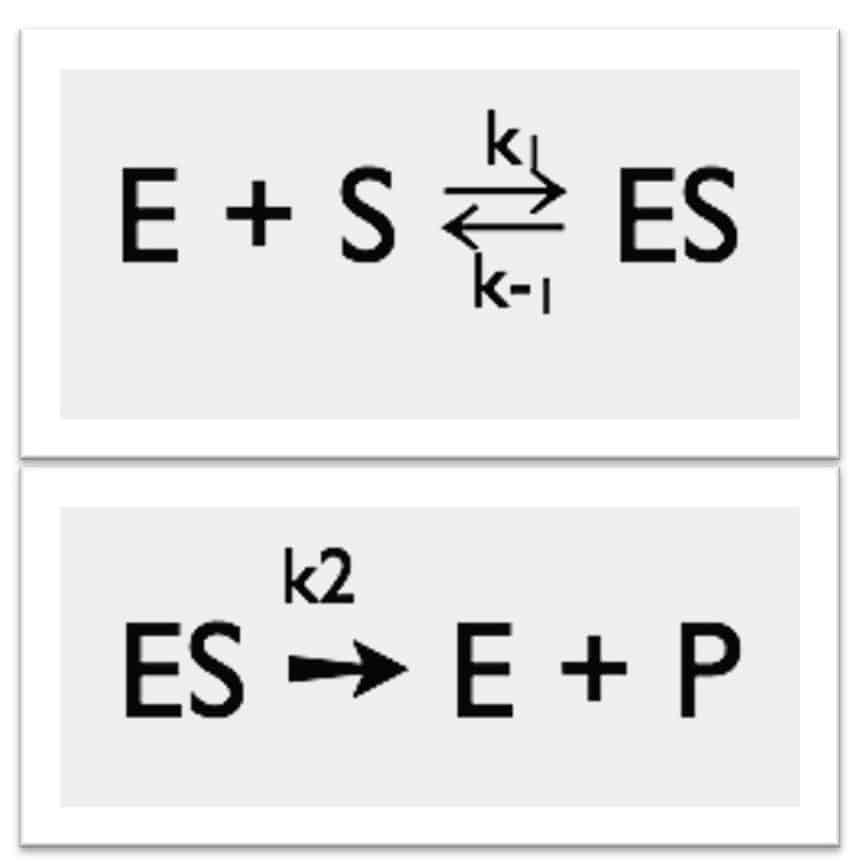
Michaelis-Menten Kinetics
- Simplest enzyme mechanism
- One reactant (S)
- One intermediate (ES)
- One product (P)
- First step: The enzyme (E) and the substrate (S) reversibly and quickly form a non-covalent ES complex.
- Second step: The ES complex undergoes a chemical transformation and dissociates to give the product (P) and enzyme (E).
v=k2[ES] - Many enzymatic reactions follow Michaelis–Menten kinetics, even though enzyme mechanisms are always more complicated than the Michaelis–Menten model.
- For real enzymatic reactions use kcat instead of k2.
Kcat
- Kcat = Rate constant of rate-limiting step
- Constant kcat is a first-order rate constant & hence has units of reciprocal time (/min or /sec)
FREE Download FullEnzyme Kinetics Notes + Exclusive Flow Chart
Or Fill Up the Form Below:
Download MORE CSIR NET Notes Here
To learn more about :
- Lineweaver-Burk-Plot (double reciprocal plot)
- Eadie–Hofstee diagram
- Hanes–Woolf plot
- Difference between them
- MM Equation Data Analysis
Join Our New CSIR NET Batch – CSIR NET + GATE JUNOON Batch 2022 starts from 1st July 2022.
Start your CSIR NET & GATE Journey With Biotecnika
New Batch Starting on 1st July 2022
Enroll in Junoon Batch & Get a Virtual internship in Cancer Genomics free
You were born to win. But to be a winner, you must plan to win, prepare to win, and expect to win.
Success in CSIR NET Exam is now just a click away – With BioTecNika’s CSIR NET Coaching you can become the next Topper of CSIR NET.
Taking only 16 Students Per batch- Enroll Today!
CSIR NET Enzyme Kinetics Notes