Rani Therapeutics in News
Rani Therapeutics has completed the initial human safety study of its pill designed to provide injectable therapies from inside the digestive tract.
Rani Therapeutics has raised $142 million to bring it with its thought poised to potentially reshape markets dominated by blockbuster injectable biologics across a variety of signs, which can be painful or difficult to work with on a regular basis, to this stage.
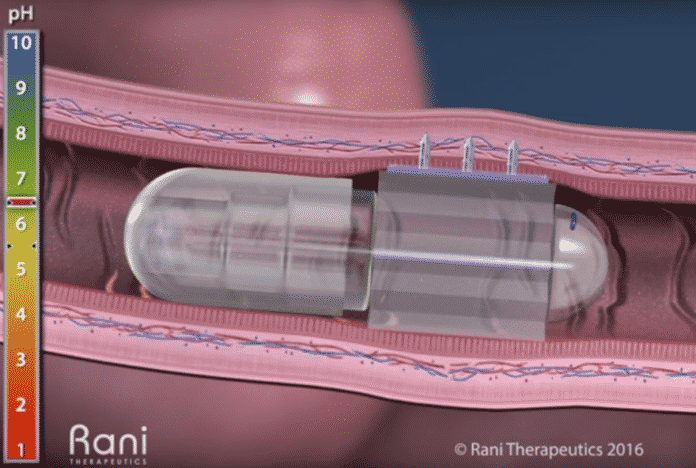
Upon entering the small intestine, then the RaniPill capsule inflates a tiny balloon, and that pushes drug-loaded needles to the wall of the organ, for fast uptake into the blood. The capsule uses other components that cannot be absorbed or passed from the body or springs, or no metal.
From the analysis, no sensations were reported since the pill inflated and deployed, and participants were able to pass the remnants of this unit within one to four times with no adverse events, the Rani Therapeutics spokesperson said. The analysis also revealed instances in both fed and fasted participants to episode, demonstrating that the existence of food does not impact performance.
Based on Rand, the first-in-human-security trial follows over 100 animal research analyzing the equivalence of the capsule’s capability to supply different injectable medications, including antibodies, peptides, and proteins. In pigs and dogs, the pill managed to provide the equivalent of 3 mg of this medication, with besting or bioavailability fitting subcutaneous injections.
The San Jose, California-based Rani Therapeutics plans to launch another research this season, comprising needles full of octreotide as well as diarrhea associated with a few tumors.
About Rani Therapeutics: The concept for Rani was developed by Mir Imran, a medical inventor, and entrepreneur who has founded more than life-sciences companies. Rani was established as a business in 2012 as a research project the multi-disciplinary life sciences laboratory of Mir, in InCube Labs.
See Also: Latest Biotech News of 2019