Nanoparticles With Crucial Role in Cancer Discovered, Dubbed Exomeres
Tumor metastasis is a multistep process that includes stimulation of angiogenesis, intravasation of tumor cells into blood or lymphatic vessels, and subsequent engraftment and extravasation to peripheral tissues where secondary tumors are seeded.
But the various intrinsic and extrinsic factors involved as well as their roles and their implications for development of therapeutic interventions to arrest metastasis is little understood. One of these factors is the newly identified nanoparticle messenger that shuttles molecules such as proteins, fats, and nucleic acids between cells.
Dubbed “Exomere”, the nanoparticle is believed to play a role in cancer development or how well patients tolerate chemotherapy.
“We found that exomeres are the most predominant particle secreted by cancer cells,” claims senior author David Lyden, Professor in Pediatric Cardiology, and a scientist in the Sandra and Edward Meyer Cancer Center and the Gale and Ira Drukier Institute for Children’s Health at Weill Cornell
Medicine. “They are smaller and structurally and functionally distinct from exosomes. Exomeres largely fuse with cells in the bone marrow and liver, where they can alter immune function and metabolism of drugs. The latter finding may explain why many cancer patients are unable to tolerate even small doses of chemotherapy due to toxicity.”The heterogeneity of exosomal populations has hindered our understanding of their biogenesis, molecular composition, biodistribution and functions.
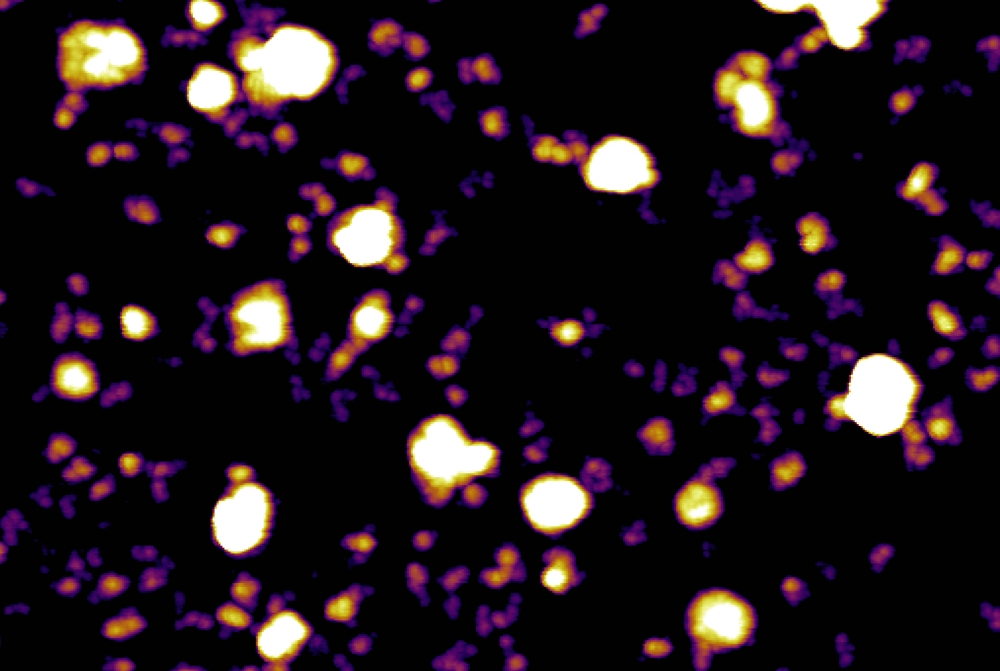
Scientists in order to elucidate the structure and subsequent details pertaining to these particles, used a cutting-edge technique called asymmetric flow field-flow fractionation (AF4) to sort exosomes that are secreted by cancer cells and contain DNA, RNA, fats and proteins.
This technology, in turn allowed the investigators to separate two distinct exosome subtypes and discover a new nanoparticle, which they named exomeres.
“Cancer is truly a systemic disease that requires multi-organ involvement to progress,” Dr. Lyden emphasized. “Our finding that tumor cells secrete these three distinct nanoparticles, that then target cells in different organs reflects this important aspect of the disease.”
Exomeres clock in at less than 50 nanometers in diameter, compared with small exosomes (Exo-S), which range from 60 to 80 nanometers in diameter, and large exosomes (Exo-L), which are 90 to 120 nanometers in diameter. “Exosomes and exomeres also have different biophysical characteristics, such as stiffness and electric charge, that likely affect their behavior in the body,” said lead author Dr. Haiying Zhang, an assistant professor of cell and developmental biology in pediatrics at Weill Cornell Medicine. “The more rigid the particle, the easier it is likely taken up by cells, rendering exomeres, which are stiffer than exosomes, the more effective messengers of transferring tumor information to recipient cells.”
The two nanoparticles are also found to influence cancer differently. Exomeres, through the study were found to target the liver to “reprogram” its metabolic function to favor tumor progression. Exomeres also carry blood-clotting factors to the liver, where they may prohibit the liver’s normal function in regulating clotting. By contrast, the study suggests that Exo-L may promote metastasis to lymph nodes, while Exo-S may support distant metastasis. Exosomes and exomeres are also detectable in bodily fluids such as lymphatic fluid, which might allow development of biomarkers for early detection of cancer or other pathological conditions.
The Cornell researchers furthering the study, intend to dig into how these different types of messengers develop, exactly what molecules each of them carry, and what their functions are at their target organ sites. “Understanding these characteristics may help scientists better understand how exomeres and exosomes help cancers grow and spread to other organs, as well as what role they may play in other diseases,” Dr. Zhang said.
“Based on our findings, the next phase will be to measure exosomes and exomeres in plasma samples to help predict organs that may be targeted for metastasis during tumor progression,” said Dr. Lyden. “This will help us better understand the biology of cancer, guide therapeutic decisions and develop novel therapies.”
“Lastly, the technique we have pioneered will likely be a valuable tool for scientists and clinicians studying the biology of complex nanoparticle populations,” Dr. Zhang said, “and may aid in the development of diagnostic tests using them as biomarkers.”