Ultrasound and Electric Field Stimulation to Treat Chronic Wounds
A Jerusalem-based medical device company developing and marketing innovative products for increasing blood flow and pain reduction, BRH Medical, has now announced 510 (k) clearance from the FDA for its innovative BRH-A2 device.
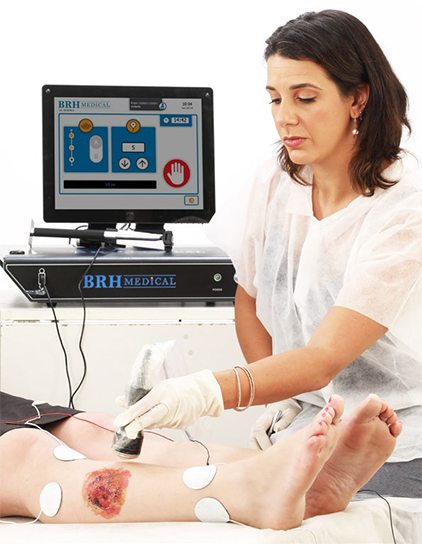
The Israel-based company’s BRH-A2 device is designed to engage and stimulate the wound from below the wound bed using both an ultrasound and electric field system which varies in frequency and intensity over the course of the treatment.
The ultrasound system is designed to work together with the electric fields to affect the tissues and muscles in the region of, and below the wound. This combination varies in frequency and intensity over the course of the treatment.
The non-invasive BRH-A2 System can be used together with any treatment modality, and its portability permits its use in clinics and hospitals. It features intuitive software for ease of use and is the only one-of-its-kind to include an integrated patient and wound management system.
“FDA clearance for the BRH-A2 represents an important milestone for our company and provides an important key to opening the huge potential of the market in the United States for our product,” said Motti Oderberg, CEO, BRH Medical. “We are now exploring the most efficient way to penetrate the market, and are currently focusing on identifying strategic partnerships,
” Oderberg added.
Ilan Feferberg, BRH Medical Chief Technology Officer and the inventor behind the newly cleared technology explained, “It has always been my desire to develop a product that would truly help cure people. Our grateful patients are proof of the effectiveness of the technology.”
Mr. Oderberg concluded, “The market for products that ease the pain of chronic wounds is large and growing. Our vision is to become the gold standard for treatment of chronic wounds of all kinds, including pressure wounds, diabetic leg wounds, and wounds caused as a result of ischemia.”