New Tissue Regeneration Method Inspired By Nature
Scientists at the University of Birmingham have now found a way of mimicking our body’s natural healing process, using cell derived nano-sized particles called vesicles, to repair damaged tissue. The team believes that the findings mark the first step in a new direction for tissue regeneration with the potential to help repair bone, teeth and cartilage.
“Though we can never fully mimic the complexity of vesicles produced by cells in nature, this work describes a new pathway harnessing natural developmental processes to facilitate hard tissue repair,” Sophie Cox, Ph.D., from the School of Chemical Engineering at the University of Birmingham, said in a statement.
The number of bone fractures is expected to double worldwide by 2020, putting added stress on healthcare systems. Osteoporosis-fragility fractures can have a significantly negative impact on quality of life.
There are significant limitations to current treatments for bone repair like autologous grafts that cannot meet demand and lead to patient morbidity, allogeneic bone lacks bioactive factors and growth factor-based approaches can lead to serious side-effects and high costs.
The new approach provides the advantages of cell-based therapies, but without using viable cells. The method harnesses the regenerative capacity of extracellular vesicles, nano-sized particles that are naturally generated in bone formation.
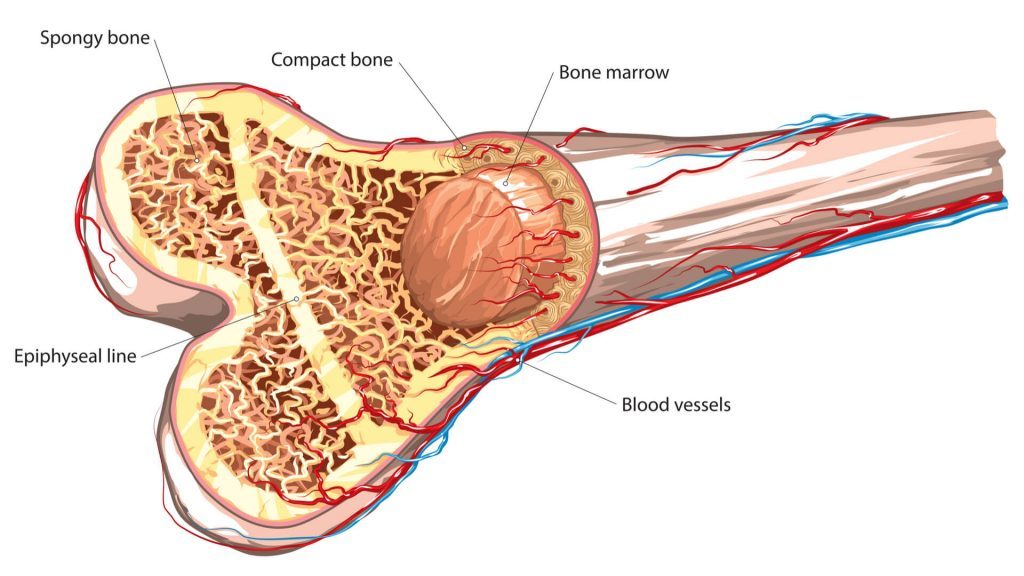
“We sought to define the capacity of EVs derived from mineralising osteoblasts (MO-EVs) to induce mineralisation in mesenchymal stem cell (MSC) cultures and delineate the underlying biochemical mechanisms involved. Strikingly, we show that the addition of MO-EVs to MSC cultures significantly (P < 0.05) enhanced the expression of alkaline phosphatase, as well as the rate and volume of mineralisation beyond the current gold-standard, BMP-2 [bone morphogenetic protein 2]. Intriguingly, these effects were only observed in the presence of an exogenous phosphate source,” wrote the investigators.
“EVs derived from non-mineralising osteoblasts (NMO-EVs) were not found to enhance mineralisation beyond the control. Comparative label-free LC-MS/MS [liquid chromatography-tandem mass spectrometry] profiling of EVs indicated that enhanced mineralisation could be attributed to the delivery of bridging collagens, primarily associated with osteoblast communication, and other non-collagenous proteins to the developing extracellular matrix. In particular, EV-associated annexin calcium channelling proteins, which form a nucleational core with the phospholipid-rich membrane and support the formation of a pre-apatitic mineral phase, which was identified using infrared spectroscopy. These findings support the role of EVs as early sites of mineral nucleation and demonstrate their value for promoting hard tissue regeneration.”
This team’s technique delivers all the advantages of cell-based therapies but without using viable cells, by harnessing the regenerative capacity of EVs that are naturally generated during bone formation, according to Sophie Cox, Ph. D., from the School of Chemical Engineering, adding that the scientists have demonstrated in vitro that if EVs are applied in combination with a simple phosphate the therapy outperforms the current gold standard, BMP-2.
If proven to be successful, patients might actually be able to see and experience their bodies recover and regenerate. It could even pave a way to help people live longer, or even indefinitely, if they are able to regenerate any tissue that gets damaged or fails, says the report.
Dr Owen Davies, EPSRC E-TERM Landscape Fellow at the University of Birmingham and Loughborough University, added, “It is early days, but the potential is there for this to transform the way we approach tissue repair. We’re now looking to produce these therapeutically valuable particles at scale and also examine their capacity to regenerate other tissues.”