Atomo HIV Self-Test Granted CE Mark by European Union
A world leader in innovative point-of-care, rapid diagnostic devices, Atomo Diagnostics, has now received CE Marking by a European Union Notified Body for its Atomo HIV Self Test.
The Australian firm is now in talks with commercialization and distribution partners to make the test widely available in Europe and other markets that recognize the CE mark.
The Atomo HIV Self Test is a lateral flow, in vitro qualitative immunoassay that detects human antibodies to HIV virus type 1 and type 2 in whole blood. It requires one finger prick of blood and can provide results in minutes, Atomo said, adding that in independent studies with untrained users, the test achieved 100 percent concordance to laboratory results.
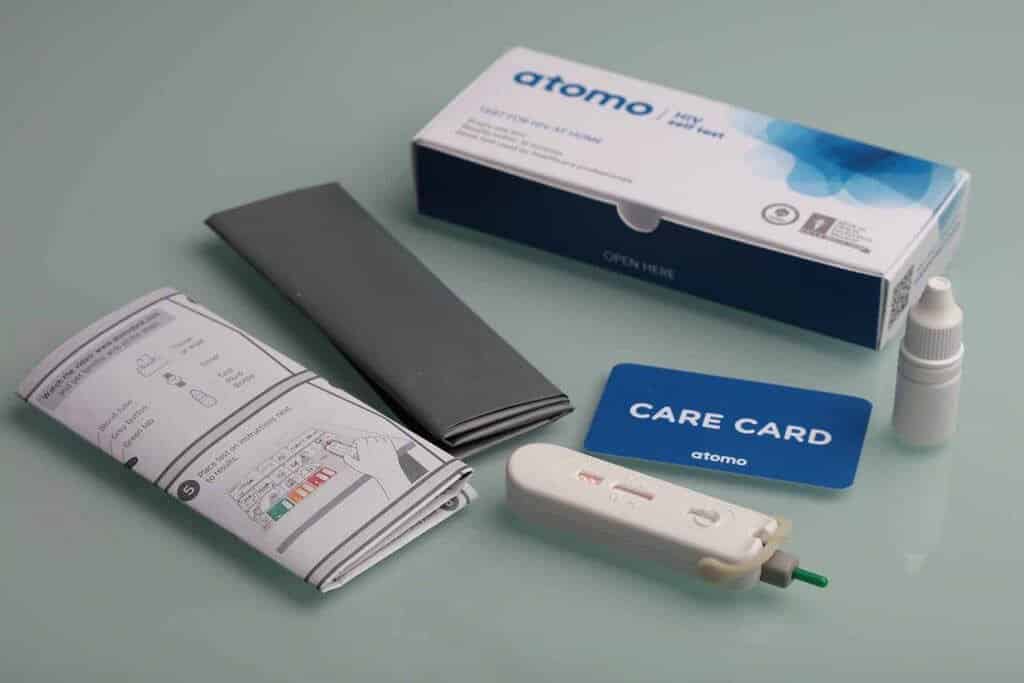
When used by untrained users in the field, the Atomo HIV Self Test demonstrated 100% concordance to laboratory results in independent studies, making it the best performing self-test approved to date. Additionally, as a 3rd generation test, the Atomo HIV Self Test can detect HIV antibodies earlier than established 2nd generation competitor tests.
“Self-testing is facilitating greater access to HIV testing for previously hard-to-reach and high-burden groups. Studies have also shown that our test has a high degree of acceptance and take-up amongst the young, which could be central to stemming infection rates and increasing access to treatment,” said John Kelly, Chief Executive Officer of Atomo Diagnostics. “CE Marking means that the Atomo HIV Self Test can now be made available to anyone who wants a safe, convenient, accurate and private way to find out their HIV status in minutes,” he added.
“HIV self-testing has the potential to be a game-changer in achieving the UNAIDS ambitious goal of 90% of all HIV positive people knowing their status by 2020,” said Linda-Gail Bekker, President of the International AIDS Society, and a member of Atomo’s Clinical Advisory Board.