Why Clinical Trials Are Going Digital and How You Can Benefit
Have you considered being a part of a clinical trial without ever visiting a hospital? Yes, the idea might seem odd to you, but this is the current reality of clinical trials in the current modern era.
Now the patients from afar wake up, put on a smartwatch that keeps the record of their vital signs, log symptoms with the help of mobile apps, and even attend the doctor’s consultation all from their home. This is an actual scenario in modern clinical research.
Clinical trails which used to be dependent on the physical site inspection and a long manual process, are now rapidly growing into digital patient-centric systems. These trends we see are the result of technological advancement we have seen in the past few years, as well as the COVID-19 pandemic, which has transformed how the trials are conducted now and, most importantly, how to build a career in this field.
What Are Digital Clinical Trials?
Digital clincal trails refers to the study where a patient can take part in the clincal trail from their home using wearable devices or simply phone apps. It utilizes electronic devices or technologies to collect data from the participants or patients and analyze them further, reducing the reliance on traditional methods and site-based procedures. This is what is known as Decentralized clinical trials (DCTs)
Key Features:
- The data is being recorded digitally instead of on paper
- Patients from far places can also participate
- Use of various apps and wearable gadgets
- Medicine is available through virtual consultation
| Tool/Technology | Purpose |
|---|---|
| eConsent | Digital informed consent |
| ePRO | Patient-reported outcomes via apps |
| Wearables | Devices to record and save data digitally |
| Telemedicine | Virtual consultation |
| EDC Systems | Data collection and management |
| CTMS | Trial planning and coordination |
Why Clinical Trials Are Going Digital
The current advancement of clinical trials towards digitalization is not just an innovation, but it also serves the purpose of giving answers to the inefficiencies faced by traditional clinical research. Let us understand in detail the reasons behind why the clinical trials are switching to digital.
- Quick recruitment and exposure to Global reach
Traditional trials often struggle with patient recruitment due to geographic limitations. It’s always been an issue in clinical trials where patients staying in remote locations are not able to participate, but with digitalization, we can overcome the following:
- Patients can be recruited from different parts of the cities or the world
- The time taken for recruitment is reduced significantly.
- To help study a broader population
A study has proven that more than half of the clinical trials are delayed due to issues faced during participation or recruitment, which, through the decentralized methods, helps solve
- Overall betterment of patient experience
The main principle behind the digital transition revolves around patient-centricity. Now, instead of continuous visits to hospitals
- Patients can participate from home
- less time taken, easy to participate
- less disruption in the day-to-day routine
- Real-Time and High-Quality Data Collection
With the help of various digital tools such as wearables and mobile apps, we can keep a continuous track of the data collected. Now, instead of collecting data at a certain interval manually, we can:
- Collection of data is done in real time
- errors caused during manual collection by humans are reduced
- helps create a structured profile for each patient
- time and cost-effective
Clinical trials are expensive and time-consuming. Demands a large amount of cost in operating the trials, including the infrastructure and various operational costs. Consume a lot of time collecting and updating the data. But through digitalization, it helps reduce the following:
- Reduce infrastructure and operational costs
- Data collection and its management are done automatically.
- The overall time taken to complete the trial is reduced.
A quick trial will ultimately lead to quicker drug development and its approvals.
- A faster pace due to the COVID-19 pandemic
The COVID-19 pandemic acted as a major catalyst for digital adoption.
- As trials were necessarily required in remote areas.
- The regulatory board oversing the all the functions gave quite a few liberties for the digital methods
- Rapid uptake of telemedicine was seen along with the increase in remote monitoring
Now, even after the pandemic, these methods and functions are not reverted but instead are being expanded to reach every corner to tackle any future crisis with ease.
Technologies Powering Digital Clinical Trials
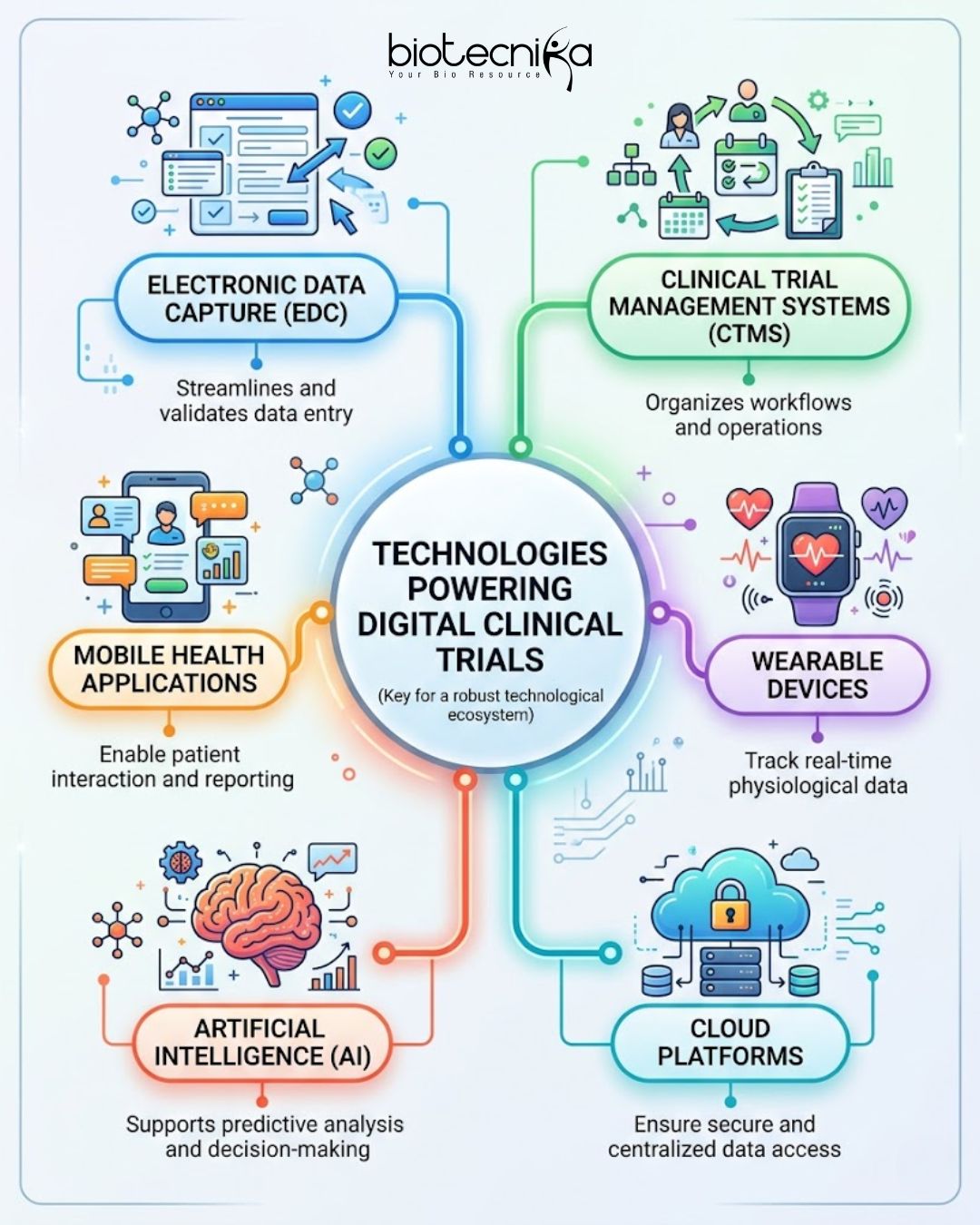
These technologies collectively enhance efficiency, scalability, and data reliability.
What are the challenges faced by digital clinical trials?
Despite their advantages, digital trials are not without limitations. Even with their immense advantages, digital trials are not without limitations. The main key challenges faced by them are as follows:
- There’s always a risk of the leaking of personal data
- There is still a lack of access to these digital tools for certain populations
- a lot more complex regulatory and compliance regulations
- It creates a dependence on the technology
- Not all trials can be fully decentralized
Due to these challenges faced by the digital clinical trials, many organizations and cross are now implementing a hybrid model, where they combine both the traditional and the digital methods for the best outcome.
Let us understand the whole concept we have covered so far through a real-world example
Consider a patient enrolled in a cardiovascular clinical trial. Let’s consider there is a patient who is enrolled in a cardiovascular clinical trial. He/she visits every week instead of visiting a hospital.
- He/She uses digital tools such as smart watches to continuously keep track of vital signs such as their heart rate.
- The collected data through the smartwatches is directly received by the researchers through the mobile app connected to it.
- Most of the consultations are conducted through virtual meetings
Why this approach is helpful:
- It provides convenient alternatives to patients living in remote places.
- collection and management of real-time data
- Helps improve overall trial efficiency.
This is the practical impact of digital transformation, not just theory but a real-world application.
How You Can Benefit from This Shift
This transformation is not only changing trials it is reshaping career opportunities in clinical research.
- Emerging Job Roles
The demand for digitally skilled professionals is increasing across the industry.
In-Demand Roles:
- Clinical Research Associate (CRA) with remote monitoring expertise
- Clinical Data Manager
- Clinical Trial Assistant (CTA)
- eClinical Specialist
- Remote Monitoring Coordinator
- High-Value Skills That Can Accelerate Your Career
To stay competitive, professionals must adapt to digital tools and workflows.
Key Skills to Focus On:
- EDC systems (e.g., Medidata Rave)
- CTMS platforms
- Trial Master File (TMF) management
- Data analysis fundamentals
- Understanding decentralized trial operations
Professionals with these skills are often preferred for modern clinical trial roles.
- Increased Remote and Global Opportunities
As trials become decentralized:
- Many roles are shifting to remote formats
- Organizations are hiring globally
- Location is becoming less of a barrier
This opens access to opportunities beyond local markets.
- Faster Career Growth and Competitive Advantage
Here is the reality; most professionals are still adapting to digital transformation. Those who learn digital tools early and understand evolving trial models gain a significant advantage in the job market. Early adopters are often the first to access better roles and faster promotions.
Future of Clinical Trials
The future of clinical research lies in integration, not replacement.
Emerging Trends:
- Hybrid clinical trials (combining site-based and remote methods)
- Increased use of artificial intelligence
- Integration of real-world data
- Greater emphasis on patient-centric designs
- Expansion of fully virtual trials in select therapeutic areas
Digital transformation is expected to continue shaping the industry in the coming years.
What does the future hold for us?
Clinical trials are evolving to meet the demands of a more connected and technology-driven world. Digital and decentralized approaches are improving efficiency, reducing costs, and enhancing patient participation.
While challenges remain, the direction of the industry is clear. For professionals in clinical research, this shift represents more than just change; it represents opportunity.
Those who adapt early, build relevant digital skills, and stay aligned with industry trends will be better positioned to grow in an increasingly competitive landscape.
As clinical trials go digital, the biggest advantage will not belong to the most experienced but to those who adapt the fastest.